
What Is Carrageenan?
Carrageenan is a hydrocolloid extracted from some red seaweeds belonging to the Eucheuma (kappaphycus), Chondrus, Gigartina and Hypnea species. It is used in a wide variety of applications in the food industry as a thickening, gelling, stabilizing and suspending agent in water and milk systems. Carrageenan is a multifunctional ingredient and it behaves differently in water and in milk systems. In water it shows typical hydrocolloid properties of thickening and gelling, while in milk systems it also has the property of reacting with proteins to furnish additional stabilizing abilities.
Carrageenan has a unique ability to form a wide variety of gel textures at room temperature: rigid or elastic, clear or turbid, tough or tender, heat stable or thermally reversible, low or high melting/gelling temperatures. It may also be used as a suspending, gelling, emulsifying, stabilizing and water retaining agent in other industrial applications.
Types of CARRAGEENAN
Carrgeenan can be classified according to its structure and chemical properties or according to its production process:
By Structure
KAPPA – rigid and brittle gel, thermo-reversible, high gel strength, showing syneresis.
IOTA – elastic gel, thermo-reversible, no syneresis, thixotropic.
LAMBDA – cold soluble, non gelling, high viscosity.
By Production process
SEMI-REFINED – opaque gel containing high cellulose and fiber levels, low degree of purity.
REFINED – clear, transparent gel, with a high degree of purity.
Different terms might be used to refer to Semi-refined carrageenan, such as PES (Processed Eucheuma Seaweed), PNG (Philippine Natural Grade) or SRC (Semi-Refined Carrageenan).
Raw Material
Carrageenan is obtained from several kinds and species of seaweeds belonging to the Rodophyceae class. The carrageenan content of commercial seaweeds varies from 20% to 40% of dry weight, depending on the seaweed species and the sea conditions, such as luminosity, nutrients, water temperature and oxygenation. Seaweeds of different species and sources produce different types of carrageenan such as kappa, iota and lambda. Some species of seaweed may produce a mixed type carrageenan such as kappa/iota, kappa/lambda or iota/lambda.
The species that produce kappa-type carrageenan are Kappaphycus Alvarezii (former Eucheuma Cottonii) and Hypnea Musciformis. The specie that produces iota-type carrageenan is Eucheuma Denticulatum (former Eucheuma Spinosum). The species that produce lambda type carrageenan generally belong to the Gigartina species. Some species like Chondrus Crispus and Gigartina skottsbergii produce mixed type carrageenan kappa/lambda or kappa/iota.
Seaweeds are usually harvested manually by fishermen in low depths at low tides or by diving using appropriate equipment. After being harvested, seaweeds are placed under the sun to dry until they reach a humidity level that is ideal for processing. Kappaphycus Alvarezii (Cottonii) and Eucheuma Denticulatum (Spinosum) seaweeds have been successfully cultivated on a commercial scale in the Philippines, Indonesia and Tanzania. Chondrus and Gigartina skottsbergii are obtained respectively from natural seaweed beds in the North Atlantic and in Chile.

Chemical Structure
Carrageenan is located in the cell wall and intercellular matrix of the seaweed plant tissue. It is a high molecular weight polysaccharide with 15% to 40% of ester-sulfate content. It is formed by alternate units of D-galactose and 3.6 anhydro-galactose (3.6-AG) joined by α-1,3 and β-1,4 –glycosidic linkage. The primary differences which influence the properties of kappa, iota and lambda carrageenan type are the number and position of ester sulfate groups as well as the content of 3.6-AG. Higher levels of ester sulfate means lower solubility temperature and lower gel strength. Kappa type carrageenan has an ester sulfate content of about 25 to 30% and a 3,6-AG content of about 28 to 35%. Iota type carrageenan has an ester sulfate content of about 28 to 30% and a 3,6-AG content of about 25 to 30%. %. Lambda type carrageenan has an ester sulfate content of about 32 to 39% and no content of 3,6-AG.
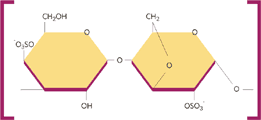
Kappa Carrageenan

Iota Carrageenan

Lambda Carrageenan
Production Process
Carrageenan may be refined or semi-refined according to the production process. Production process for semi-refined carrageenan is always the same. For refined carrageenan, there are three methods used in the industry: Drum Drying, Alcohol Precipitation, and Gel Press.
Semi Refined – Standard Method

Refined by Drum Drying

Refined by Alcohol Precipitation

Refined by Gel Press

Properties
Hot Water
All carrageenan types are soluble in hot water at temperatures above its gel melting temperature. The normal amplitude of solubility temperature is between 40º and 70º C, depending on the solution concentration and the presence of cations..
Cold Water
In cold water, only lambda-carrageenan and the sodium salts of kappa and iota carrageenan are soluble. Potassium and calcium salts from kappa and iota type carrageenan are not soluble in cold water, but will swell as a function of concentration and type of cations present as well as water temperature and condition of dispersion.
Hot Milk
All carrageenan types are soluble in hot milk but some types are strongly affected by calcium ions presence. On cooling all of these solutions tend to gel. The strength of the gel and its consistency depend on the solution concentration and the sensitivity of the carrageenan to calcium ions.
Cold Milk
Lambda carrageenan is soluble in cold milk, due to its insensitivity to the presence of potassium and calcium ions. Kappa and iota carrageenan are insoluble in cold milk, but may be used efficiently to thicken and/or gel cold milk solutions when used in conjunction with a phosphate such as tetra-sodium pyrophosphate (TSPP).
Concentrated Sugar Solutions
All carrageenan types are relatively insoluble in concentrated sugar solutions at room temperature. However, kappa and lambda carrageenan are soluble in sucrose solutions as high as 65%, at temperatures above 70ºC. Iota carrageenan is only sparingly soluble in concentrated sugar solution at any temperature.
Concentrated Salt Solutions
All carrageenan types are insoluble in concentrated salt solutions (20% to 25% of sodium chloride) at room temperature . Iota and lambda carrageenan are soluble hot. Kappa carrageenan is insoluble hot.
Gelling Mechanism
Hot aqueous solution of kappa and iota carrageenans have the ability to form thermo-reversible gels upon its cooling. This phenomenon occurs due to the formation of a double helix structure by the carrageenan polymers. At temperatures above the melting point of the gel, carrageenan polymers exist in solution as random coils. On cooling of the solution, a three-dimensional polymer network builds up in which double helices form the junction points of the polymer chains. Further cooling leads to aggregation of these junction points to build a three-dimensional gel structure. The presence of kinks in the chain, as well as the quantity, type and position of ester sulfate groups have important effects on gelling properties. This gelling mechanism is basic for kappa and iota carrageenan solutions. Calcium or potassium salts are needed to obtain water gel, but they are not necessary for milk gels.
Texture
Kappa and iota carrageenans will only gel in the presence of certain cations. Kappa type carrageenan is potassium ion sensitive and it produces rigid and brittle gels in aqueous solutions with potassium salts. Kappa carrageenan gel presents syneresis (spontaneous extrusion of water through the surface of gel when it is at rest) and the higher the potassium concentration in solution the higher the syneresis. The iota type carrageenan is calcium ion sensitive and it forms soft and elastic gels in aqueous solutions with calcium salts. Iota carrageenan does not present any syneresis. The gel strength is directly proportional to the concentration of carrageenan and salts. The use of excessive amount of potassium salts will produce a weaker gel strength. The gel formed is thermo-reversible and it may be subject to heating and cooling cycles, without a noticeable change in the gel structure (neutral pH). The gelling and/or melting temperatures depend on the concentration of cations. The increase of potassium or calcium salts concentration in aqueous carrageenan solutions will result in an increase in the gelling temperature.
The viscosity of carrageenan solutions should be determined under conditions where there are no tendencies for the solution to start gelling. When a hot carrageenan solution is cooled, the viscosity increases gradually until the gelling temperature is reached. As the gel starts to form, there is a sudden and a very substantial increase in viscosity. Therefore, the viscosity measurements of carrageenan solutions should be determined at sufficiently high temperatures (75º C) to avoid the effect of gelation. The carrageenan concentration is generally 1.5% by weight of the water solution. Commercial carrageenans are available generally in viscosities ranging from about 5 to 800 cps when measured in 1.5% solutions at 75º C. The viscosity of carrageenan solutions depends on concentration, temperature, the presence of other solutes, the type of carrageenan and its molecular weight. Viscosity increases exponentially with a higher molecular weight and/or higher solution concentration. Viscosity decreases with increase in temperature and vice versa.
Carrageenan solutions are quite stable at neutral or alkaline pHs. At lower pHs their stability decreases, especially at high temperatures. As the pH is lowered hydrolysis of the carrageenan polymer occurs, resulting in loss of viscosity and gelling capability. However, once the gel is formed, even at low pHs (3.5 to 4.0), hydrolysis no longer occurs, and the gel remains stable. For practical applications, it is important to be aware of the limitations of carrageenan under acid conditions (either solution or gel). Therefore, processing of carrageenan solutions at low pHs and high temperatures for a prolonged period of time should be avoided.
One of the most important properties that makes carrageenan different from other hydrocolloids is its ability to interact with milk proteins. The high reactivity of carrageenan with milk is due to the strong electrostatic interaction between the negatively charged ester sulfate groups in the carrageenan molecule, with strong positive charges of the milk casein micella. Another form of interaction is through links established among ester sulfate groups of carrageenan with carboxylic residues of amino acids that make up the protein. The reactivity with proteins is dependent on many factors such as concentration of carrageenan, type of protein, temperature, pH and isoelectric point of the protein. The ability of carrageenan to complex with milk proteins combined with its water gelling properties makes it an efficient ingredient for stabilizing and gelling milk products.
Kappa carrageenan shows an unusual synergism with locust beam gum (LBG) in aqueous gel systems. The interaction is marked by a considerable increase in the gel strength, an improve in water binding capacity, a change in gel texture from brittle to elastic, and a reduction in the degree of syneresis.
Iota carrageenan presents a synergism with starches. A system containing a mixture of iota carrageenan and starch presents an increase in viscosity of as much as 10 times the viscosity of starch alone. Therefore, iota-type carrageenan may be useful in altering the textural, mouthfeel and processing properties of starch based systems.
At low concentrations iota carrageenan water gels have thixotropic rheological properties. These gels may be fluidized by agitation or shear and will form elastic gels when allowed to stand at rest. This thixotropic property is particularly useful to suspend insoluble particles such as spices in salad sauces. Water gels formed with kappa-type carrageenan does not present this thixotropic property. Once the gel is broken it will not reset to its original form unless the gel is heated and cooled again.
Functionality and Applications
The uses of carrageenan are concentrated in the food industry. Carrageenan applications are generally divided into milk based systems, water based systems and beverages. However, there are many other applications for carrageenan in a large variety of industrial applications. Carrageenan has many functions according to its uses and applications: gelling, thickening, emulsion stabilizing, protein stabilizing, particle suspension, viscosity control and water retention are just a few.
Food Industry
Milk Products

Ice creams, Chocolate milk, Flans, Puddings, Whipped cream, Yogurts, Creamy milk desserts, Cheeses, Dry mix desserts, Coconut milk
Confectionary

Dessert Gel, Jam, Marmalades, Marshmallows, Gum drops, Confitures, Meringues
Meat and Fish Products

Cooked ham, Imitation meat, Sausage, Canned meat, Hamburger, Pureed Meat, Poultry, Processed Meat, Kanikama
Beverages

Clarifying and refining of juices, beers, wines and vinegars; Syrups, Powdered fruit juices, Diet shakes
Bakery Products

Cake icings, Tart fillings, Bread dough, cake glaze
Dressings and Soups

Dry mix salad dressings, Powdered soups, Mustard, White sauce, Ready to eat sauces
Other industrial applications




Tooth paste, Air fresheners, Pet food , Cosmetics, Paints, Emulsions, Vegan capsules
Specifications
General Properties
| Appearance | Yellowish powder |
| Particle size | Mesh 100/200 |
| Moisture content | Max. 12 % |
| Water absorption | Max. 75 c.c. |
| Total ash | Max. 15 % |
| Foreign insoluble material | Máx 1,0 % |
| PH (sol 1.5 % at 20 º C) | 8,0 to 11,0 |
| Gel Strength (water sol 1.5 % at 20 º C) | 100 to 1200 g/cm2 |
| Viscosity (water sol 1.5 % at 75º C) | 30 to 300 cps |
| Melting Point | 50 to 70º C |
| Setting Point | 30 to 50º C |
| Solubility | Water at 85ºC |
Microbial Properties
| Total Plate Count | < 5000 CFU / g |
| E.Coli | Negative |
| Salmonella | Negative |
